Chlorobutanol
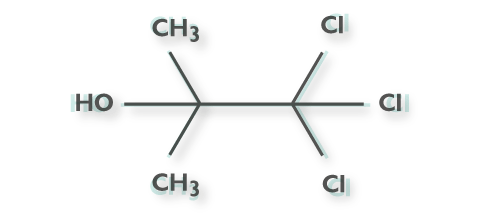
Chlorobutanol
Chlorobutanol is a well accepted widely used, very effective preservative in many pharmaceuticals and cosmetic products, e.g. injections, ointments, products for eyes, ears and nose, dental preparations, etc. It has antibacterial and antifungal properties and has been used for more than 130 years when it was first manufactured. Chlorobutanol is typically used at a concentration of 0.5 % where it lends long term stability to multi-ingredient formulations.
Chlorobutanol is manufactured in a modern plant certified as both EN ISO 9001 and EN ISO 14001 compliant. The plant has been certified by local authority as ”operating under strict c-GMP conditions“ and has passed rigorous on-site audits by many of the leading pharmaceutical manufacturers of the world.
Chlorbutol (6640-h)
Chlorbutol (BAN)
Acetone-Chloroforme; Alcohol Trichlorisobutylicus; Chlorbutanol; Chlorbutanolum;Chloretone;
Chlorobutanol (rlNN); Chlorobutanolum; Trichlorbutanolum.
1,1,1- Trichloro-2-methyl-2-propanol.
Chemical formula
Hemihydrate: C4H7CI3O ·1/2 H2O, Anhydrous: C4H7CI3O
Formula Weight
Hemihydrate: 186.47, Anhydrous: 177.46
CAS —57-15-8 (chlorobutanol anhydrous): 6001-64-5 (chlorobutanol hemihydrate).
Pharmacopoeias.In Chin., Cz., Egypt., Hung., Ind., Nord., and Yug. (all with 1/2 H2O). In Braz., Mex., and U.S.N.F. (anhydrous or with 1/2 H2O). Aust., Br., Eur., Fr., Ger., Gr., It., Neth., and Swiss have separate monographs for anhydrous and hemihydrate on permits up to 6% of water. Turk. has anhydrous.
Description:
Colourless or white crystals or a white crystalline powder with a characteristic somewhat camphoraceous odour. It sublimes readily.
Solubility:
B.P. solubilities for both forms are: slightly soluble in water; soluble in 0.6 of alcohol; freely soluble in chloroform; very soluble in ether; soluble in glycerol (85%). U.S.N.F. solubilities also for both forms are soluble 1 in 125 of water, 1 in 1 of alcohol, and 1 in 1 10 of glycerol; freely soluble in ether; in chloroform, and in volatile oils.
Melting Point:
Anhydrous Approx. 207ºF (97ºC), Hemihydrate Approx. 169ºF (76ºC)
Stability
This product is stable at ambient temperatures and atmospheric pressures for a period of 5 years (Hemihydrate) or 3 years (Anhydrous) according to ICH - conditions. Avoid prolonged exposure to air. The anhydrous grade is hygroscopic. Hazardous polymerisation is not expected to occur.
Uses and Administration
Chlorobutanol has antibacterial and fungicidal effects and it is used at a concentration of 0,5% as a preservative in injections, eye drops and mouth washs, salves, creams and ointments as well as cosmetics.
Chlorobutanol is manufactured as Hemihydrate and Anhydrous. The anhydrous form is useful in formulations where the absence of water is desired.
Toxicology
Chlorobutanol is widely used as a preservative in a number of pharmaceutical formulations, particularly ophthalmic preparations. Although animal studies have suggested that Chlorobutanol may be harmful to the eye, in practice the widespread use of Chlorobutanol as a preservative in ophthalmic preparations has been associated with few reports of adverse reactions. The incidence of toxicity is low. The acute oral LD50 is 510 mg/kg in rats.
Skin contact:
The acute dermal LD50 is greater than 2000 mg/kg in rabbits. A single dermal application of 2000 mg/kg produced 10% mortality but no signs of toxicity. Topical application may cause mild anesthesia of the skin. Local effects included a mild moderate erythema (redness). Rarely, sensitization reactions may occur in previously exposed individuals. Non-irritant to rabbit skin following a 24 hour exposure.
Eye contact:
Non-irritant to rabbit eyes.
Storage Requirements:
Containers should be stored in a cool, dry, well-ventilated area away from flammable materials and sources of heat or flame. Store away from food stuffs or animal feed. Exercise due caution to prevent damage to or leakage from the container. Avoid prolonged exposure to air.
Other informations:
For the process of manufacturing of chlorobutanol no animal raw materials are used.
Chlorobutanol has no allergic potential.
Proprietary Names
Multi-ingredient preparations.
Acne-Ban, Aezodent, Alodont, Alsol, Angicola, Auralgicin, Aurisan, Balsamorhinol, Bezotol, Biphédrine Aqueuse, Blue Gel, Cerumenol, Cerumol, Centanorm, Chlorbutone, Ciella, Coliquifilm, D.D.D., Dermidex, Eludril, E.D.P.-Evans Dermal Powder, Emoform, Fialetta Odontalgica, Dr. Knapp, Frador, Givalex, Karvol, Monphytol, Nazophyl, Otocaina, Otocerum, Otomidone, Otorrilan, Paf., Pernomal, Pueriotine, Sedaural, Stovédrine, Vitasédine, Wax Aid. Preparation details are given in Part 3.
From: MARTINDALE-The Extra Pharmacopoeia, Thirtieth Edition
References Cited:
1. Yousef RT, et al. Effect of some pharmaceutical materials on the bactericidal activities of preservatives. Can J Pharm Sci 1973; 8: 54-6.
2. FriesenWT, Plein EM. The antibacterial sability of chlorobutanol stored in polyethylene bottles. Am J Hosp Pharm 1971; 28: 507-12.
3. Holdsworth DG, et al. Fate of chlorbutol during storage polyetylene dropper containers and simulated patient use. J Clin Hosp Pharm 1984; 9: 29-39.
4. Richardson NE, et al. The interaction of preservatives with polyhydroxy-ethylmethacrylate (polyHEMA) J Pharm Pharmacol 1978; 30: 469-75.
5. Anonymous. Handbook of pharmaceutical excipients. Washington and London:
American Pharmaceutical Society of Great Britain, 1986.
6. Nair AD, Lach JL. The kinetics of degradion of chlorobutanol.
J Am Pharm Assoc (Sci) 1959; 48: 390-5.
7. Patwa NV, Huyck CL. Stability of chlorobutanol. J Am Pharm Assoc 1966 NS6: 372-3.
8. Hawley, Gessner G., The Condensed Chemical Dictionary, 10th Edition. Van Nostrand Reinhold Company. NY, pp. 238, 235, 1981.
9. DIMDI: RTECS (RT00)
Unsere Produkte
- SIE & ER® LipCare
- Peru-Lenicet®
- Essitol®
- Aluminiumacetat-Tartrat Lösung DAB 10
- Alsol®
- Lenicet®
- Aluminiumacetate-Tartrate
- Chlorobutanol
MSDS download
CHLOROBUTANOL x ½ H2O
English version
CHLOROBUTANOL x ½ H2O
German version
CHLOROBUTANOL anhydrous
English version
CHLOROBUTANOL anhydrous
German version
